Prof. WU Dayu of Changzhou University, a user of the China's Steady High Magnetic Field Facility (SHMFF) of the Hefei Institutes of Physical Science of the Chinese Academy of Sciences, together with his collaborators, proposed a simple mechanical strategy to optimize the electronic structures of the catalytic center by mechanically induced spin transition, and developed a new method for designing efficient biomimetic catalysts.
Results were published in Angewandte Chemie International Edition.
In recent years, the synthesis of transition-metal catalysts has received considerable attention. However, to improve catalytic activity/selectivity, it's imperative to precisely control the electronic structure of the catalytic center at the atomic level.
Wu's team in-situ induced partial spin crossover (SCO) of the iron catalytic active center around the grid-like lattice from high spin (HS, S = 5/2) to low spin (LS, S = 1/2). Due to the partial SCO of the iron center, the mixed-spin (MS) nanosheet catalyst exhibited a high CO yield of 19.7 mmol g-1 and a selectivity of 91.6%, much higher than its high-spin counterpart.
In this study, SHMFF experimental conditions were used to confirm the spin transition of the catalytic active center.
Density functional theory (DFT) calculations showed that the electron configuration of the low-spin three-dimensional (3d) orbitals effectively increased the overlap of bonding orbitals between O-2p and Fe-3dxy/dyz, which significantly promoted the selective adsorption of CO2. However, the high-spin 3d orbital overlapped with the O-2p orbital through the 3dz2 anti-bonding orbital, which greatly weakened the bonding interaction between the catalyst and the substrate, and reduced the catalytic activity.
In addition, DFT calculations showed that the low-spin state played a key role in reducing the activation barrier. The researchers used electron paramagnetic resonance spectrometer, Mossbauer, magnetic susceptibility and other experimental methods to analyze and characterize the electronic structure of catalysts before and after mechanical exfoliation.
Aside from that, they calculated the physicochemical mechanism behind spin catalysis by DFT calculation.
This study lays the foundation for the development of highly active, low-cost and environmentally friendly CO2 reduction catalysts, and further provided an important guarantee for solving the current energy and environmental crisis and achieving the goal of dual carbon.
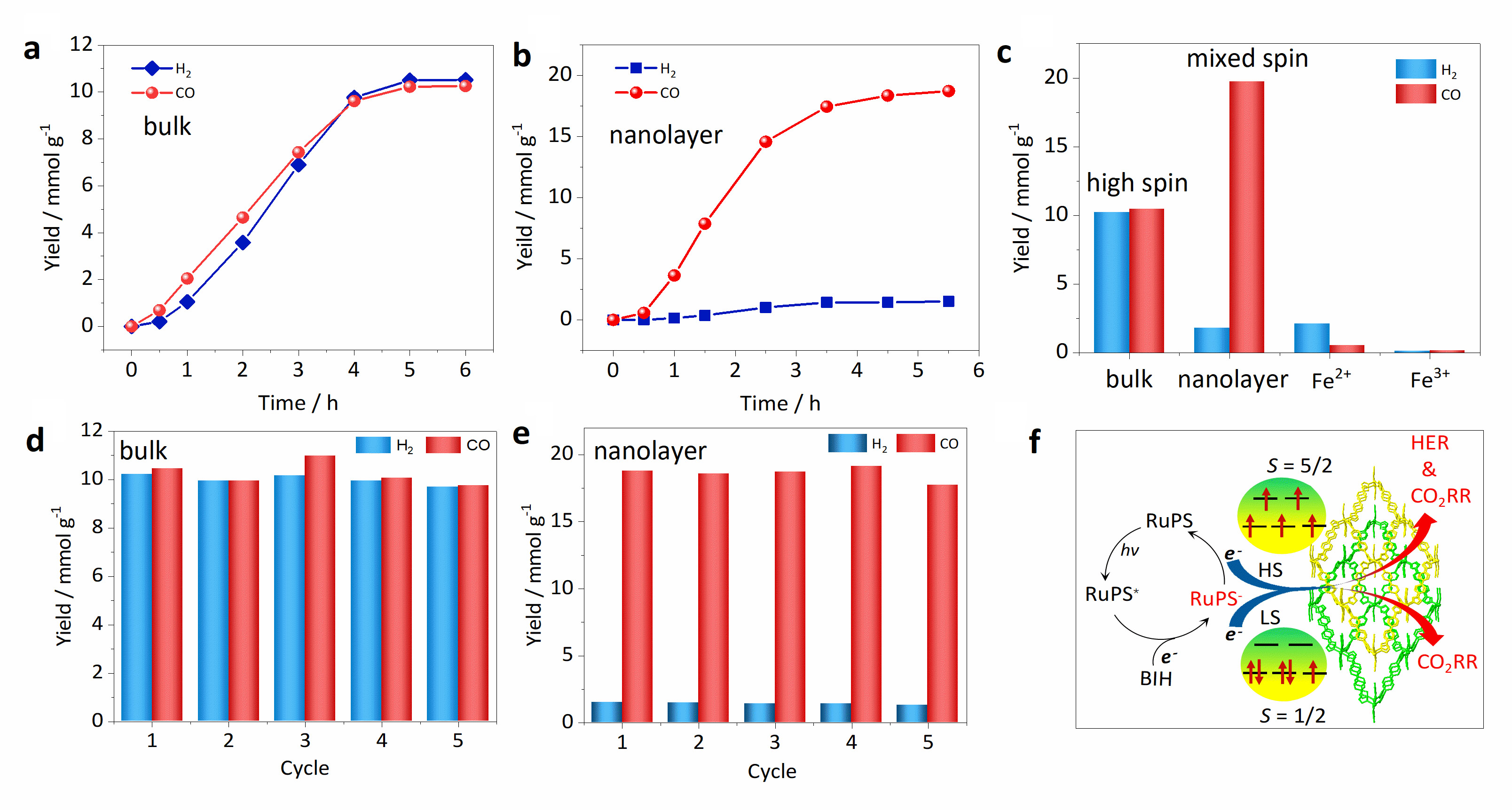
Catalytic performance. Time-dependent H2 and CO yield with (a) HS bulk and (b) MS nanolayer o-1. (c) H2 and CO yields through HS bulk o-1, MS nanolayer and control samples as catalysts after four h irradiation. Recycle experiments for H2 and CO production with (d) HS bulk o-1 and (e) MS nanolayer o-1 as catalysts. (f) The proposed photocatalytic mechanism in this work. (Image by WU Dayu)






