A research group led by Prof. WANG Junhu from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) has proposed a new strategy to effectively regulate the reaction pathway of zinc-ferrum double oxide Fenton-like catalyst through visible light irradiation, which can help to modulate the mechanism from radical to non-radical of heterogeneous catalyst in Fenton-like reaction.
The study was published in Chemical Engineering Journal on Nov. 8.
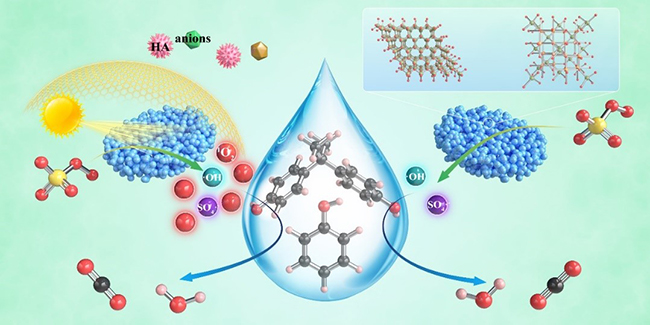
Possible mechanisms of organic pollutant degradation in ZFO-4/PMS/vis system (left) and ZFO-4/PMS system (Image by ZHANG Liang)
The quenching of radical groups in Fenton-like reactions by various inorganic anions or high concentration of organic matter limits their value in industrial applications.
Non-radical-dominated systems can effectively overcome these limitations, showing high activity against the degradation of contaminants under widespread matrix interference in water.
In this study, the researchers prepared a series of Zn-Fe double oxides by calcining the Zn1-xFex-Fe Prussian blue analogues in an ambient atmosphere. They revealed that the samples ZFO-1 and -4 were composed of nanocomposited ZnFe2O4 and ZnO, while ZFO-2 and -3 were consisted of ZnFe2O4 and Fe2O3.
Under the co-existence of visible light irradiation and peroxonosulfate (PMS), Zn-Fe double oxides showed good catalytic performance for the oxidation of various organic pollutants with the leaching of trace iron, and superior pollutant decomposition efficiency in high concentration organic matter humic acid, varous kinds of anions and simulated actual water system. This suggested that visible light plays an important role in the PMS-based Fenton-like reaction over Zn-Fe double oxides.
Through radical quenching experiments and EPR spectroscopy characterization, photoinduced electrons and holes could be employed to trigger efficient activation of PMS, thus transforming the reaction pathway in the prepared Zn-Fe dioxides from radical pathway to non-radical pathway dominated by singlet oxygen (1O2).
"This work provides a new perspective for using solar energy to regulate radical and non-radical pathways in advanced PMS-based oxidation processes," said Prof. WANG.






