A "Matrix Biology" paper by the Werner group (IMHS) shows how wound healing gene expression signatures are expressed in tumors and can be used to predict cancer outcome. The paper establishes a functional link between wound fibroblasts and cancer-associated fibroblasts through expression of certain extracellular matrix genes.
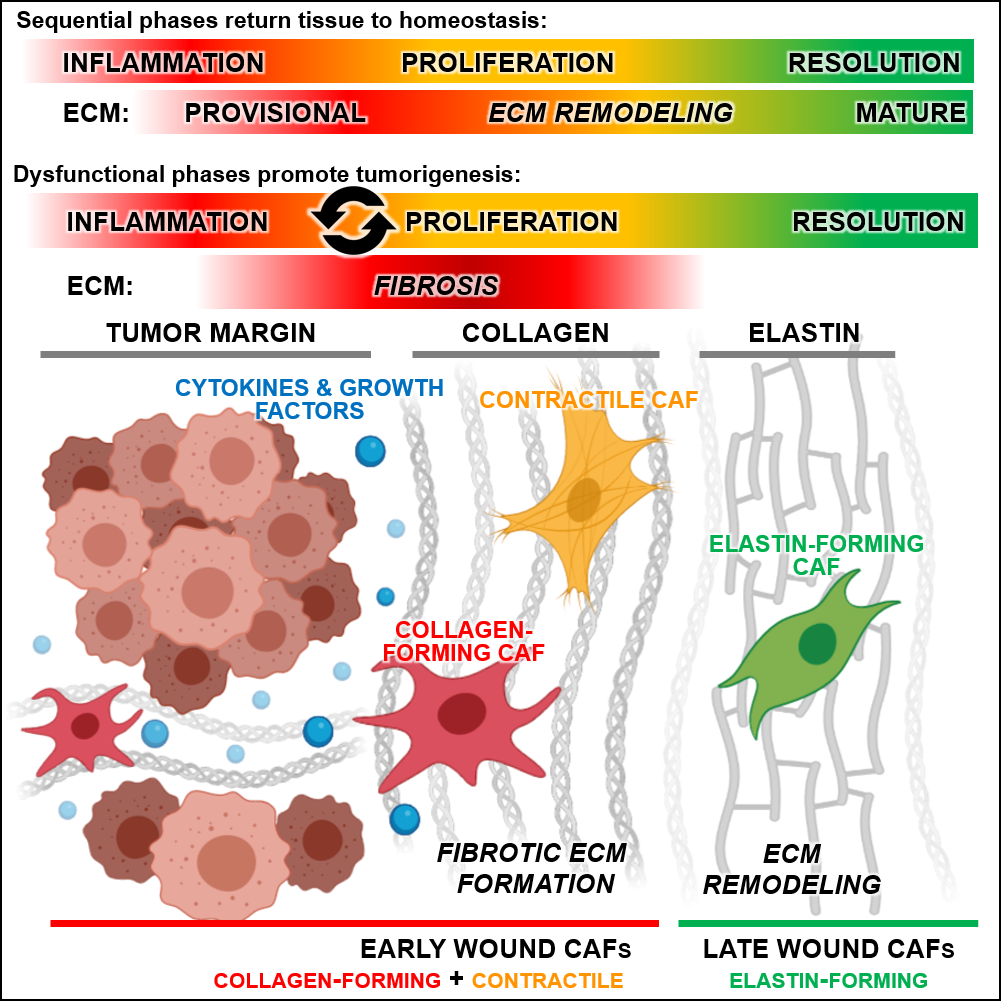
Wound healing and cancer formation share many cellular and molecular parallels, but the two have not been systematically compared. Using transcriptomics at the whole tissue, single-cell and spatial levels, Wietecha et al. profiled skin wounds across time and compared them to tumors. They discovered prognostic healing gene signatures expressed in various cancers that link wound fibroblasts to a common feature of malignant tumors mediated by cancer-associated fibroblasts - fibrosis.
The healing signatures from the early and late wounds enriched for divergent extracellular matrix repertoires that were visualized in primary melanoma biopsies and predicted cancer survival and recurrence. The authors further showed how the early wound and cancer-associated fibroblast subtype is activated by the RUNX2 transcription factor. The results pave the way for the development of innovative healing-based diagnostic and prognostic tools to better identify tumor spread and more accurately predict cancer outcome.
Link to the publication in external page"Matrix Biology"






