Article courtesy of Brookhaven National Laboratory, by Ariana Manglaviti
Quantum materials display exotic behaviors due to the effects of quantum mechanics, or how matter acts on the very small scale of atoms and subatomic particles. The technologically relevant properties of quantum materials result from complex interactions of electron charge, orbital, and spin and their coupling to the material's crystal structure. For example, in some materials, electrons can flow freely without any resistance; this phenomenon, called superconductivity, could be harnessed to transmit power more efficiently. Typically, these properties emerge at low temperature, where crystals display low (broken) structural symmetry.
"Not surprisingly, this low-temperature regime is well studied," said Emil Bozin, a physicist in the X-ray Scattering Group of the Condensed Matter Physics and Materials Science (CMPMS) Division at the U.S. Department of Energy's (DOE) Brookhaven National Laboratory. "Meanwhile, the high-temperature regime remains largely unexplored because it's associated with relatively high symmetry, which is considered uninteresting."
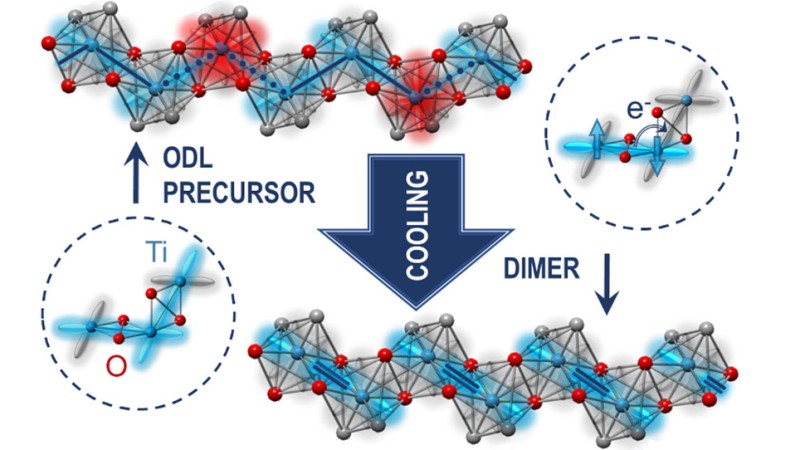
But Bozin and colleagues have recently discovered states of local symmetry breaking at high temperature. These local states are associated with electronic orbitals (regions within an atom where electrons are most likely to be found) that serve as orbital degeneracy lifting (ODL) "precursors" to what happens at low temperature. Orbital degeneracy refers to when orbitals have the same energy. Lifting of this degeneracy means that some orbitals will have a relatively higher energy and others a lower energy.
"We think such local states are in some way enablers of the material properties of interest that materialize at much-lower temperature," explained Bozin.

Top row: Robert Koch, Ryan Sinclair, Marshall McDonnell, Runze Yu. Middle row: Milinda Abeykoon, Matthew Tucker, Alexei Tsvelik, Simon Billinge.
Bottom row: Haidong Zhou, Weiguo Yin, and Emil Bozin.
The scientists first observed these local states in 2019 in a material (copper iridium sulfide) with a metal-insulator transition and in an iron-based superconductor. Now, the team-representing Brookhaven Lab; DOE's Oak Ridge National Laboratory; University of Tennessee, Knoxville; and Columbia University-has found them in an insulator containing sodium, titanium, silicon, and oxygen. This insulating material is one of the minerals forming the Earth's upper mantle. Beyond the geological interest, it's a candidate for quantum spin liquids (QSLs), an exotic state of matter in which electron spins remain fluid-like to the lowest temperatures, constantly fluctuating. QSLs could provide a material platform for quantum computing, spintronics (electronics based on electron spin rather than charge), superconductivity, and other technologies.
"Our findings suggest that this ODL precursor behavior at high temperature may be quite common and should be considered in theoretical studies to understand the functional properties of quantum materials," said physicist Weiguo Yin of the CMPMS Division Condensed Matter Theory Group.
To probe the material's atomic structure, the team analyzed how the material scattered neutrons and x-rays. Both probes are needed because of their different sensitivities to particular elements based on atomic weight. Unlike x-rays, neutrons can distinguish light elements, like oxygen. With the neutron and x-ray scattering patterns, the local arrangement of atoms can be deduced through the atomic pair distribution function (PDF), which describes the distances between different atoms in a sample. Using software, scientists can then find the structural model that best fits the experimental atomic PDF function.
This work was supported by the DOE Office of Science. The neutron scattering experiments were performed at the Nanoscale-Ordered Materials Diffractometer (NOMAD) beamline (BL-1B) of the Spallation Neutron Source (SNS) at Oak Ridge and the x-ray scattering experiments at the PDF beamline (28-ID-1) of Brookhaven's National Synchrotron Light Source II (NSLS-II). Both NSLS-II and SNS are DOE Office of Science User Facilities.






