Joint Press Release by Hokkaido University and Sapporo Medical University.
Researchers have revealed the modulatory effect of the anti-inflammatory metabolite itaconate on T helper and T regulatory cells, which may lead to new therapeutic approaches to treating some autoimmune diseases.
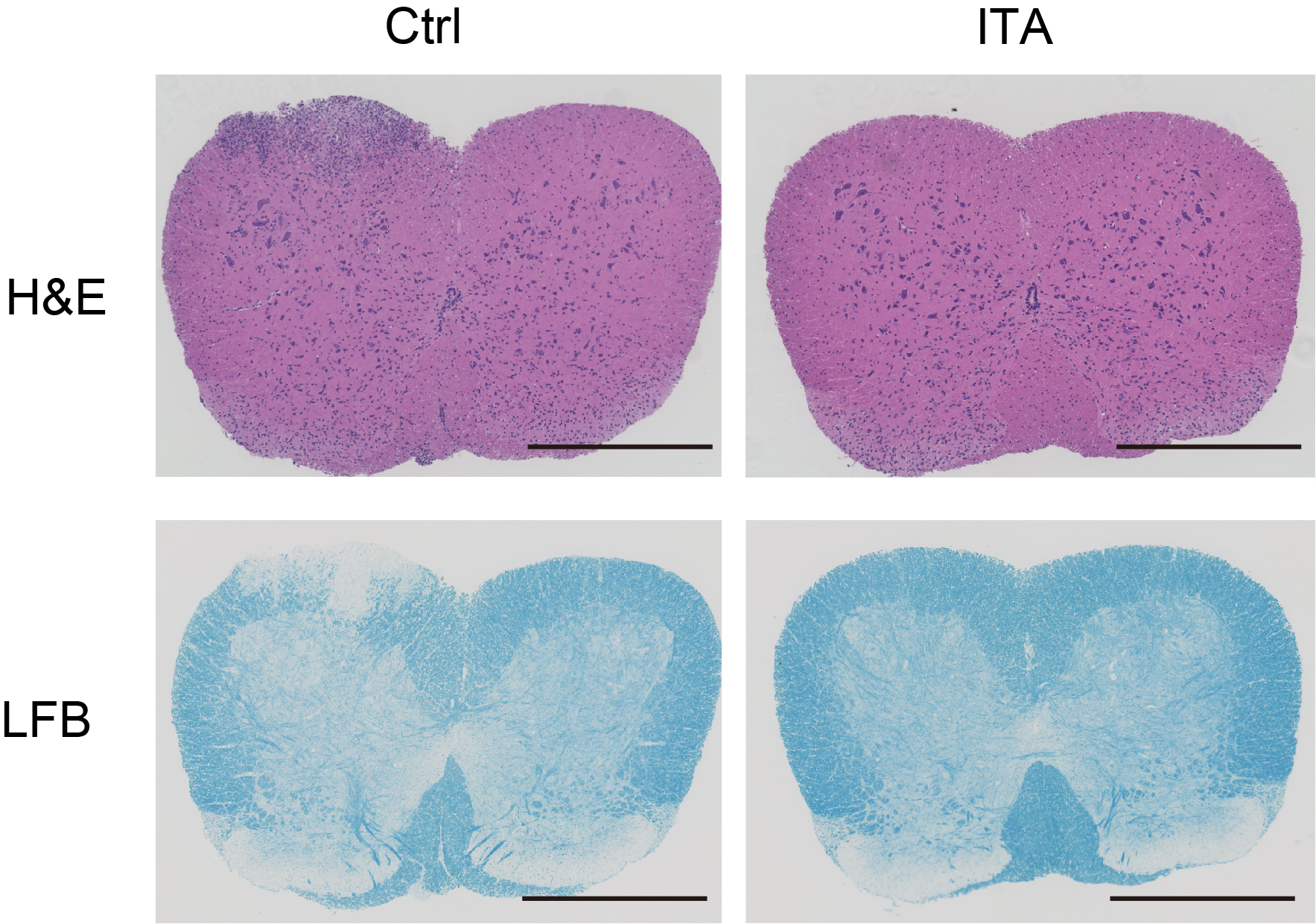
In mice models with adoptive transfer experimental autoimmune encephalomyelitis, treatment with itaconate (right) greatly ameliorates the effects of the disease, compared to untreated mice (Kuniyuki Aso, et al. Nature Communications. February 27, 2023).
Autoimmune diseases occur when the immune system attacks its own body. There are more than eighty known types of autoimmune diseases. In many cases, autoimmune diseases can be treated by suppressing the immune system; however, a side effect of such treatment is that the patient has an increased risk of severe infectious diseases, which is a leading cause of death. Hence there is a need to establish novel therapies for autoimmune diseases to reduce the risk of infectious diseases.
A research team led by Professor Tatsuya Atsumi, Assistant Professor Michihito Kono and graduate student Kuniyuki Aso at Hokkaido University, along with Senior Lecturer Masatoshi Kanda at Sapporo Medical University, has studied the effect of the molecule itaconate on the immune system. Their findings, which have implications for treating autoimmune disorders, were published in the journal Nature Communications.
"Multiple sclerosis (MS) and systemic lupus erythematosus are two of the many autoimmune diseases caused by a dysregulation of T cells," Kono explained. "We were interested in two types of T cells: T helper 17 (Th17) and regulatory T (Treg) cells. These cells have the same origin but have opposite functions in autoimmune diseases, and cell metabolites modulate their action. The metabolite we focused on was itaconate (ITA), as it has been shown to have anti-inflammatory, antiviral, and antimicrobial effects."
The researchers showed that, in cell cultures, ITA inhibited the differentiation of Th17 cells which have the potential to elaborate autoimmune diseases, and promoted that of Treg cells, which can ameliorate them. Further, in mice models with experimental autoimmune encephalomyelitis, ITA reduced the disease symptoms. Further tests were conducted to confirm that this effect was due to its effect on T cells.
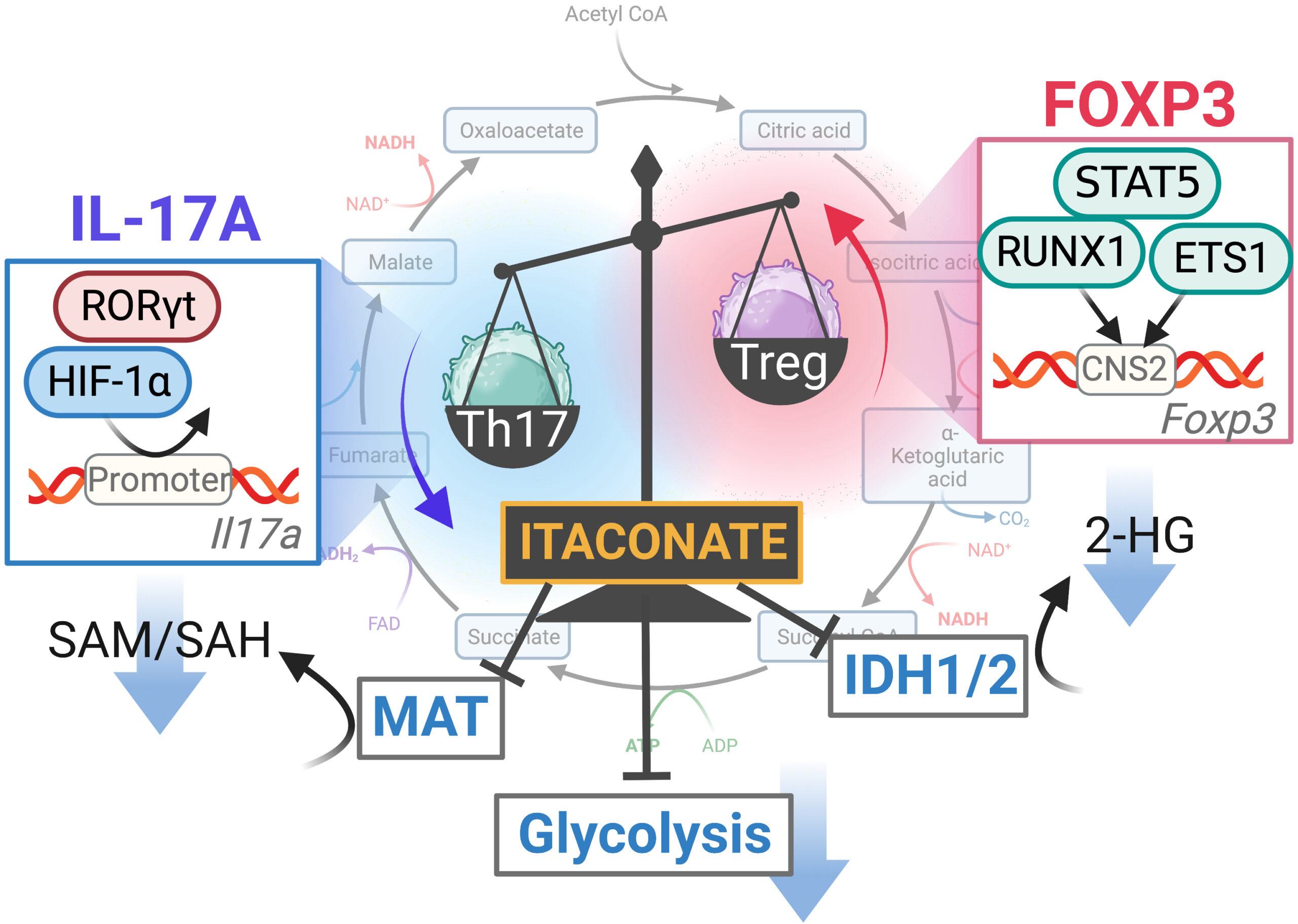
Itaconate (ITA) inhibits the differentiation of Th17 cells and promotes the differentiation of Treg cells by inhibiting glycolysis, methionine adenosyltransferase (MAT) and isocitrate dehydrogenase (IDH1/2), which in influence expression of the cytokine IL-17A and the regulator protein FOXP3 (Michihito Kono).
Investigations into the mechanism of action of ITA revealed that it inhibits essential metabolic pathways, glycolysis, oxidative phosphorylation, and methionine metabolism in Th17 and Treg cells. "ITA inhibits these pathways by directly inhibiting the enzymes methionine adenosyltransferase and isocitrate dehydrogenase, resulting in change of S-adenosyl-L-methionine/S-adenosylhomocysteine ratio and 2-hydroxyglutarate levels," Kono elaborated. "The altered cell metabolites also indirectly affect the chromatin accessibility of essential transcription factors and the synthesis of proteins required for the differentiation of Th17 and Treg cells."
"Our results explain the mechanisms that underlie the modulation of T cell differentiation," he concluded. "This could eventually lead to simple therapeutic approaches which regulate T cell differentiation, thereby treating T cell-mediated autoimmune diseases."
Original Article:
Kuniyuki Aso, et al. Itaconate ameliorates autoimmunity by modulating T cell imbalance via metabolic and epigenetic reprogramming. Nature Communications. February 27, 2023.
DOI: 10.1038/s41467-023-36594-x
Funding:
This study was supported by the Japan Agency for Medical Research and Development (AMED; JP20ek0410078h); Grants-in-Aid for Regional R&D Proposal-Based Program from Northern Advancement Center for Science & Technology of Hokkaido Japan; the Kurata Grants from the Hitachi Global Foundation; Tokyo Biochemical Research Foundation; and Inamori Foundation.






