Long-term and excessive use of antibiotics has caused the spread of antibiotic resistance. The time- and cost-consuming process of new antibiotic development results in the much slower emergence of new antibacterial drugs than that of bacterial resistance. The emergence of superbugs has been growing to become a dominant challenge in human health. Tigecycline is regarded as the last line of defense to combat multidrug-resistant Klebsiella pneumoniae. However, increasing utilization has led to rising drug resistance and treatment failure. Tigecycline resistance of Klebsiella pneumoniae remains a global problem that urgently needs to be solved.
On June 29, 2022, Prof. DU Yongzhong from College of Pharmaceutical Sciences, Zhejiang University, Prof. LU Xiaoyang and JIANG Saiping from The First Affiliated Hospital, Zhejiang University School of Medicine co-published an article entitled TPGS-based and S-thanatin functionalized nanorods for overcoming drug resistance in Klebsiella pneumonia in the journal Nature communications. Their research demonstrates that D-alpha tocopheryl polyethylene glycol succinate(TPGS)-modified and S-thanatin peptide (Ts)-functionalized nanorods based on calcium phosphate nanoparticles can overcome tigecycline resistance of Klebsiella pneumonia.
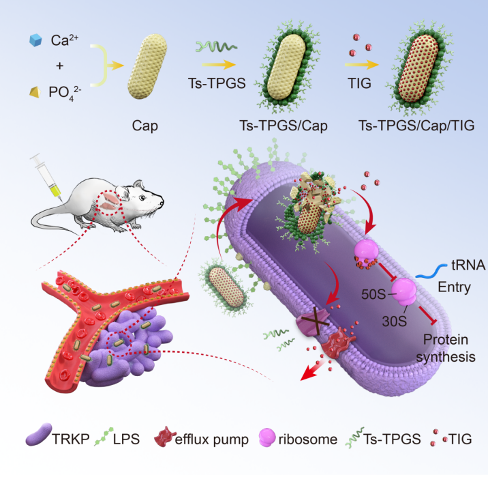
Fig. 1 Schematic depiction of the fabrication of the nanorods and the mechanism of overcoming the tigecycline resistance of TRKP. TIG: tigecycline; Ts-TPGS/Cap/TIG: tigecycline-loaded TPGS and S-thanatin functionalized calcium phosphate nanorods; TRKP: tigecycline-resistant Klebsiella pneumonia. LPS: lipopolysaccharide.
Firstly, the researchers prepared the tigecycline-loaded TPGS-modified and S-thanatin peptide-functionalized nanorods, Ts-TPGS/Cap/TIG (TTCT), and characterized the properties of TTCT. They found that the prepared Ts-TPGS/Cap nanorods could effectively encapsulate TIG and achieve sustained drug release. The TTCT with particle size of ~25 nm would not disassemble in circulation and displayed excellent stability at room temperature.
Secondly, the researchers evaluated the antibacterial activity of TTCT and explored the underlying mechanisms of overcoming resistance. They found that Ts-TPGS/Cap exhibited targeting and enhanced accumulation in both Klebsiella pneumonia (KPN) and TRKP through the binding between Ts and LPS. TPGS could exert its inhibitory capacity on the activity of efflux pumps and the expression of acrA, acrB and ramA in TRKP. In this way, the TIG concentration inside bacteria was significantly higher in the TTCT group than other groups. The synergistic antibacterial capacity between Ts and TIG further enhanced the antibacterial activity TTCT, thus overcoming the drug resistance of TRKP.
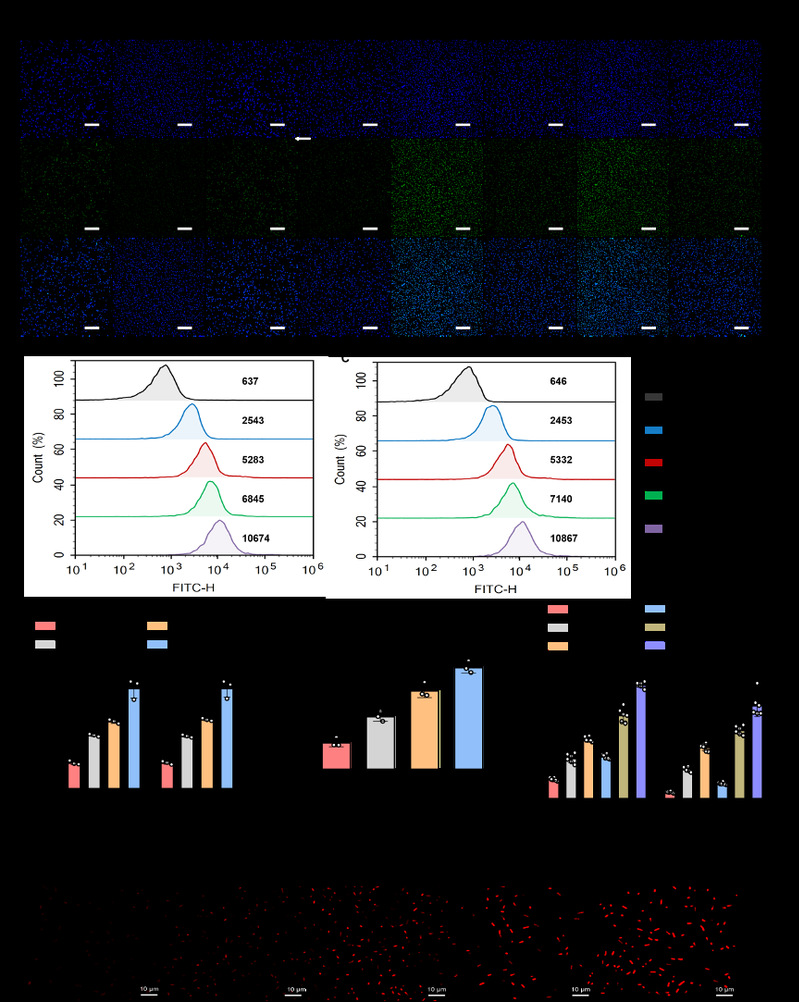 Fig. 2 Investigation of the mechanisms by which TTCT overcomes drug resistance.
Fig. 2 Investigation of the mechanisms by which TTCT overcomes drug resistance.
In mice with pneumonia, Ts-TPGS/Cap specifically accumulated in the lungs. TTCT administration could significantly reduce the white blood cells and neutrophil counts in blood samples, and decrease the total cell and C reactive protein (CRP) levels in bronchoalveolar lavage fluid (BALF). Moreover, TTCT was capable of ameliorating neutrophil infiltration in the lungs and reducing bacterial colonies from BALF, thus apparently increasing the survival rates of mice with pneumonia caused by TRKP.
Overall, a TPGS-based and Ts-modified nanodrug delivery system was designed. The prepared nanorods can enhance tigecycline accumulation in bacteria via the inhibitory effect on efflux pumps exerted by TPGS and the targeting capacity of S-thanatin to bacteria. The synergistic antibacterial capacity between S-thanatin and tigecycline further enhances the antibacterial activity, thus overcoming the tigecycline resistance of TRKP. The findings provide a therapeutic strategy for infections diseases caused by MDR gram-negative bacteria.
Source: The First Affiliated Hospital, Zhejiang University School of Medicine
Photo credit: the research team of DU Yongzhong, LU Xiaoyang and JIANG Saipin