
Left to right: MIT researchers Pranam Chatterjee, Shuguang Zhang, and Rui Qing
Photo courtesy of MIT Media Lab.
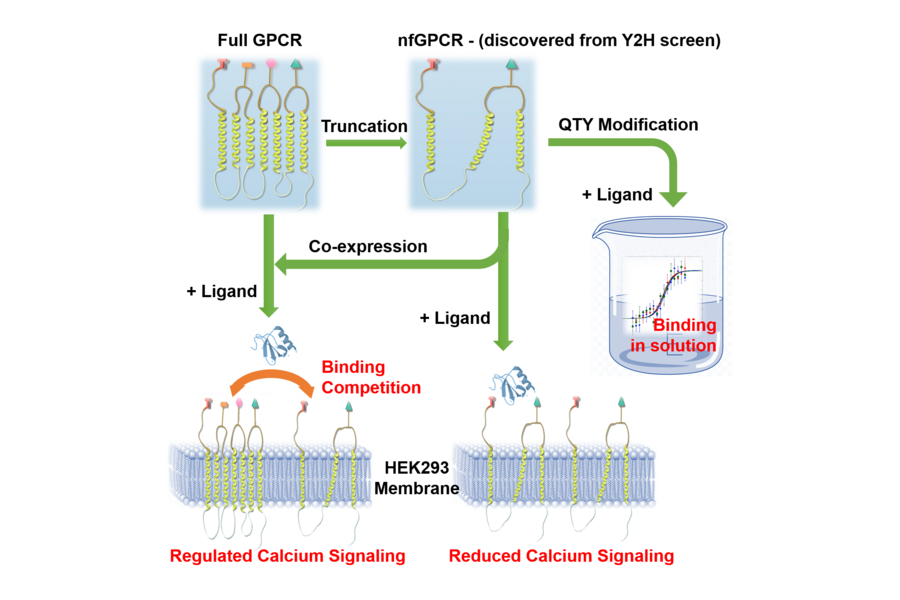
A schematic illustration of the of the unexpected discovery of non-full-length G protein-coupled receptors and their function in either native (left) or soluble (right) forms.
Photo courtesy of MIT Media Lab.
MIT Media Lab researchers have discovered that shortened versions of immunity-related protein receptors, long written off as incomplete and therefore nonfunctional, can bind with their natural counterparts on the cell membrane and may represent pathways to new medications, therapies, and bionic body parts.
Although chemokine receptors with truncated structure have previously been considered biologically inconsequential, a paper published in Cell Press's iScience journal reports that truncated versions of two such receptors, CXCR4 and CCR5, bind with their corresponding ligands and play a role in protein functionality.
"This unexpected discovery that short receptors can recognize their respective ligands potentially opens a new vista into a new regulation system in biology," says Shuguang Zhang, a principal research scientist and head of the the Laboratory of Molecular Architecture in the Media Lab's Molecular Machine group, and who is a senior author of the paper. "It will likely stimulate researchers to ask questions that have been overlooked and to venture beyond current paradigms to discover, characterize, and design proteins, and develop new technologies and medicine."
Zhang uses an everyday analogy to describe the binding capability of the shortened receptors.
"The receptor is like our hand holding a teacup," he says. "You don't need five fingers to hold it. Two fingers are good enough."
The truncated receptor proteins in their native form are non-water-soluble, and to maintain their structure require detergents that make studying them difficult. In order to avoid detergents, the researchers had to convert the receptor proteins into water-soluble forms by substituting hydrophobic amino acids with structurally similar hydrophilic ones using Zhang's invention known as QTY code. This process allowed the researchers to study the physiological and functional properties of the chemokine receptors without detergent in vitro. The receptors were also reconverted to a non-water-soluble form to be observed in vivo.
Not only could the reconverted, native variants of the truncated receptors be expressed and position themselves on the cell membrane to interact with their respective ligands, they demonstrated a regulatory effect on full-length receptors, blocking them from reaching the cell surface. Zhang points out that this phenomenon needs further study and experimentation but explains how it could potentially lead to therapies to thwart diseases such as cancer.
"Cancer cells often have a malfunction in their cell signaling process. In many cases, they over- express G protein-coupled receptors including chemokine receptors CXCR4 and CCR5, which we report on in the current study. The truncated but functional receptors could interfere with cancer cell signaling," says Zhang.
Regulating full-length receptors' binding with surface proteins could also potentially impede the entrance of a virus, and in so doing reduce or prevent infection.
Zhang and his colleagues are also developing water-soluble decoy receptors that are the functional equivalents of native receptors to "soak up" ligands, which could result in reducing metastasis. A previous publication explained how such a technique could also diminish cytokine storms, an excessive and damaging immune response that can occur in patients with Covid-19 and other viral or bacterial infections, including HIV and hepatitis.
"Combining those results and this current truncated receptor study, we aim to have more control on the structure and functionality of the potential system we are developing," says Zhang.
Meanwhile, the water-soluble versions of the truncated receptors could potentially provide an abundant nanomaterial ingredient for use in bionic sensing devices. Unlike native membrane proteins, which are almost impossible to use for the production of the several-kilogram quantities of nanomaterials required for such devices, minimal water-soluble proteins can be chemically synthesized much more easily.
"If the short receptors work as well as the long ones - and even at about 50-80 percent as well - you can make the nanomaterials by chemical synthesis to easily scale up production," says Rui Qing, a Koch Institute for Integrative Cancer Research scientist who, along with fellow researchers Fei Tao and Pranam Chatterjee, is a lead author on the iScience paper.
Also, the smaller size of the truncated receptors would help them work well as micro-scale, very specific electronic sensing devices, says Qing.
"It's always better to have a smaller molecule on the surface to not attenuate the signal," he says.
The observation of the shortened but functional CXCR4 and CCR5 raises many questions, Zhang says, such as what the smallest functional receptors are that can exist in living organisms and whether there are DNA sequences that specifically code for non-full-length receptors in all genomes. This discovery also raises questions about "pseudogenes," which are truncated genes, and how many such pseudogenes have really been systematically studied at the protein level in cells and in organisms.
As Zhang points out, unexpected discoveries have resulted in many major breakthroughs in science, such as the discovery of the role of microRNA, which was once overlooked and is now recognized as indispensable to genetic regulation, especially in highly evolved biological systems.
"Unexpected discoveries often shed light into our knowledge blind spot," says Zhang. "The most important scientific implication is to stimulate people to ask questions about what we have missed or overlooked, peel another layer of the knowledge onion to look into the complexity of life. Unexpected discoveries often open up many possible ways to develop new technologies and many applications. They force people to think again."






