Type 2 diabetes (T2DM) is a systematic multi-organ metabolic disease, which is characterized by dynamic interplay among different organs.
Pancreatic islet-liver axis is closely associated with normal glucose regulation and homeostasis maintenance. The dysfunction of the interplay between these organs may lead to T2DM progression.
Recently, a group led by Prof. QIN Jianhua from the Dalian Institute of Chemical Physics (DICP) of the Chinese Academy of Sciences (CAS) developed a multi-organoid system from human induced pluripotent stem cells (hiPSCs) to simulate human liver-islet axis in normal and T2DM.
This study was published in Advanced Science on Dec. 23, 2021.
"Stem cell-derived organoid, a new class of 3D tissues, has the key structural and functional features of in vivo counterparts," said Prof. QIN.
This newly developed multi-organoid system could simulate human liver-pancreatic islet axis in physiological and pathological conditions, and enable 3D co-culture of hiPSC-derived liver and islet organoids for up to 30 days.
The researchers found that the generated liver and islet organoids exhibited favorable growth and improved tissue-specific functions in this perfused microfluidic 3D culture system.
"The sensitive glucose-stimulated insulin secretion was from the islet organoids and it increased glucose utilization in the liver organoids," said Prof. QIN. This reflected the cooperative interaction between the two organs in this perfused co-culture system.
Transcriptional analyses revealed that the activated signaling pathways were associated with glucose/CYP450 metabolism and glycolysis/gluconeogenesis in liver and islet organoids.
What's more, this microfluidic islet-liver organoid system exhibited mitochondrial dysfunction and decreased glucose transport capacity under hyperglycemic condition, which might be attributed to the alleviation by treatment with anti-diabetic drug.
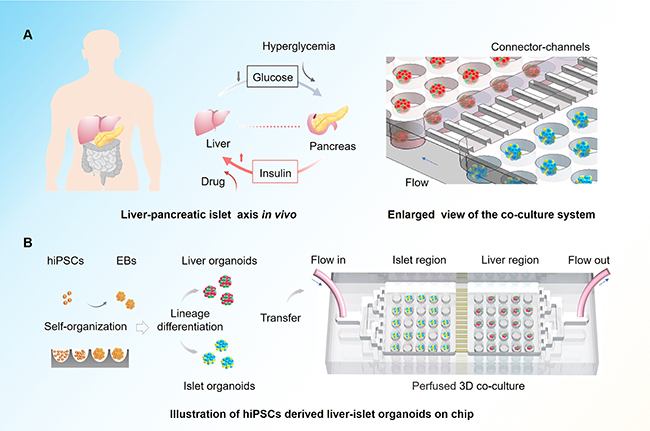
"This novel system can not only reflect the feedback loop within the liver-islet axis, but also exhibit relevant responses to high glucose and anti-diabetes drug, which is not easily achieved by conventional cell culture and animal models," said Prof. QIN. "It will provide a unique platform for the study of complex T2DM pathogenesis and drug development."
This research was supported by the National Key R&D Program of China and the Strategic Priority Research Program of CAS.






