Fifteen years ago, doctors in Europe noticed a remarkable thing happening in people with chronic hepatitis B infections. When patients went off their medications, the virus started to come back - and then some of the patients were cured. Something was goading their immune systems to go after the virus as the infection rebounded.
Now, a UC San Francisco team has uncovered how this works: a certain type of T cell, some of which coordinate immune responses, spots the infection in the liver and mobilizes an attack. Without these immune cells, which are called CD4+ T cells, the infection continues. The finding points out new avenues to achieve a cure.
"It's taken us many years to explain why some of our patients are able to beat hepatitis B," said Jody Baron , MD, PhD, a professor of Medicine at UCSF and co-senior author of the paper, published April 29 in Science Translational Medicine. "We think this could lead to much better treatments based on the liver's natural biology."
Up to 640,000 Americans and 250 million people worldwide live with chronic hepatitis B, which spreads via bodily fluids from an infected person and is usually transmitted at birth from a mother to a child. The disease raises the risk of liver failure, liver cancer, and serious scarring of the liver called cirrhosis. Vaccines and therapies can hold the infection at bay, but more than 1 million people die each year from the virus.
Hepatitis B often becomes chronic when infection occurs early in life, before the immune system can mount an effective response. By contrast, newly infected adults are far more likely to clear the virus.
How childhood hepatitis B becomes chronic
Baron's team set out to understand what triggers this immune response - and why it succeeds in some chronic hepatitis B patients but not others.
"When treatment stops in a structured way, about a third of patients can mount the right immune response and clear the virus - something we almost never see during treatment," said Stewart Cooper, MD, a member of the UCSF Liver Center and co-senior author of the paper. "But that observation didn't lead to change for most patients."
To understand, the researchers turned to the lab. They engineered mice to make the hepatitis B virus or its proteins, and to lack immune cells from birth. Then, they gave the mice a transplant of fresh immune cells, testing whether these immune cells could identify the hepatitis B virus as a threat.
In adult mice, immune transplants containing CD4+ T cells quickly detected the hepatitis B proteins and set off an immune alarm. But it didn't work in young mice: the CD4+ cells didn't react.
"The mouse models show how childhood hepatitis B becomes chronic, while adult hepatitis B can be overcome," said Gabriela Fragiadakis , PhD, a computational immunologist and professor at UCSF and co-senior author of the paper.
A UCSF-led team discovered that immune-cell clusters in the liver, drawn in by signals from CD4 immune cells (green), can beat hepatitis B in the adult mouse liver (left), but not in the young mouse liver (right). The same thing occurred in a subset of adult patients with chronic hepatitis B who managed to beat the infection when they were taken off antiviral medication. Images by Baron Laboratory, UCSF
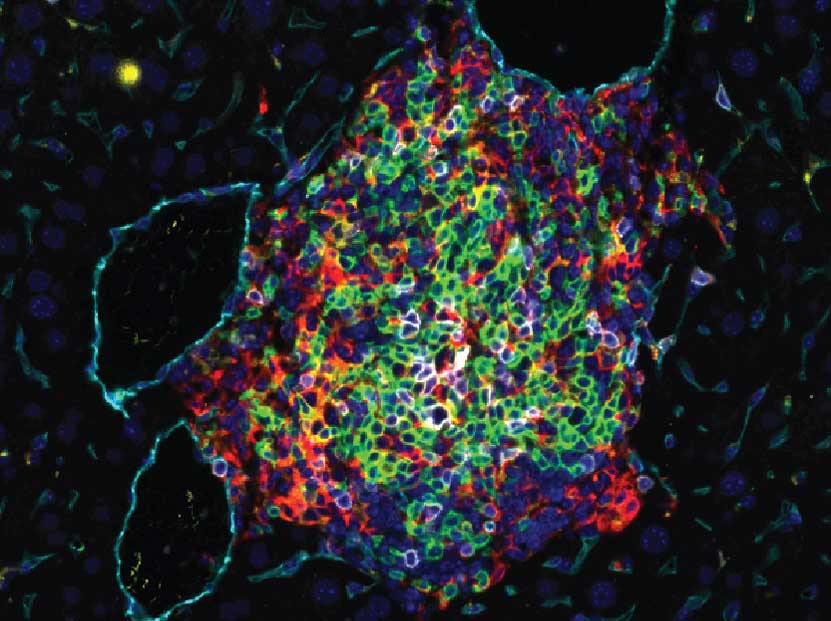
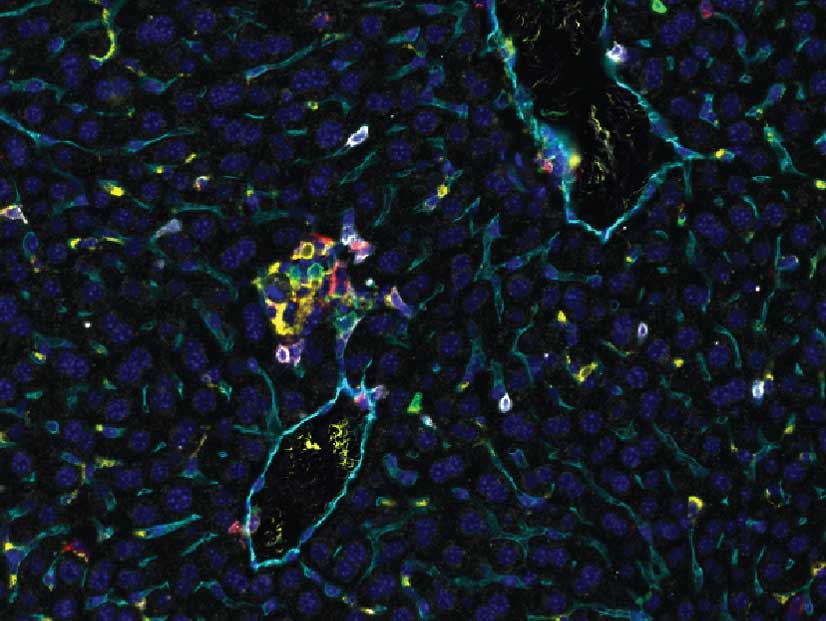
New results suggest CD4+, not CD8+, immune cells are key
Next, the group analyzed blood samples from patients who had been treated for chronic hepatitis B. The patients had been carefully weaned off their antivirals, but only some of them were able to kick the infection.
In patients who ultimately cleared the virus, CD4+ cells in the liver became more active as the virus replicated. This did not happen to the CD4+ cells in patients who failed to clear the virus. These findings challenge the long-standing focus on CD8+ "killer" T cells as the drivers of hepatitis B clearance.
"Seeing the same immune patterns in both the mouse model and in patients gives us confidence we're capturing something real about how this disease works," Fragiadakis said.
If CD4+ cells are required for clearance, therapies could be designed to activate them as patients come off antivirals, prompting the immune system to finish the job.
Authors: Other UCSF authors are Jillian M. Jespersen, PhD; Lia Avanesyan, MS; Jean Publicover, PhD; Nicholas D. Carey; Ravi K. Patel, PhD; Austin W. Edwards; Sarah Stenske; Jae Shin, MD, PhD; Jiajing Li, PhD; Margaret Simone; Nayvin W. Chew; Nicholas Wong, JD; Suprita Trilok, MS; Arjun Rao, PhD; Christopher P. Loo, PhD; Monika Sarkar, MD, MAS; Alexis J. Combes, PhD; and Mark R. Segal, PhD. For all authors see the paper.
Funding: The National Institutes of Health (P30DK026743, P30DK063720, S10OD026940-01, 75N93020D00005, R01AI139762, R01DK103735, 5F31DK112607, T32DK060414, F31DK135386, T32AI007334); Ibrahim El-Hefni Liver Biorepository; UCSF Liver Center; The California Liver Institute; Norman Raab Foundation; Technical Training Foundation; Ing Foundation; UCSF Data Science CoLab; Disease to Biology CoLab; UCSF Parnassus Flow Cytometry CoLab; and the UCSF Bakar ImmunoX Initiative.
About UCSF: The University of California, San Francisco (UCSF) is exclusively focused on the health sciences and is dedicated to promoting health worldwide through advanced biomedical research, graduate-level education in the life sciences and health professions, and excellence in patient care. UCSF Health , which serves as UCSF's primary academic medical center, includes top-ranked specialty hospitals






