A joint research team from Nitto Boseki Co., Ltd. (Nittobo) and Tohoku University has revealed that Poly(ionic liquid)s (PILs) can achieve high carbon dioxide (CO₂) adsorption when their counter anions are exchanged. This discovery provides a critical new design guideline for the development of high-performance CO2 recovery devices and gas separation membranes.
The research was led by Associate Professor Kouki Oka of the Institute of Multidisciplinary Research for Advanced Materials, Tohoku University, with the results published online in the chemical engineering journal Reaction Chemistry & Engineering on March 9, 2026.
PILs are known for their strong ability to attract CO₂ and for their stability as solid materials. However, conventional anion exchange methods struggle to remove inorganic salts, which are by-products of the manufacturing process. These impurities make it difficult to accurately evaluate the materials' true performance.
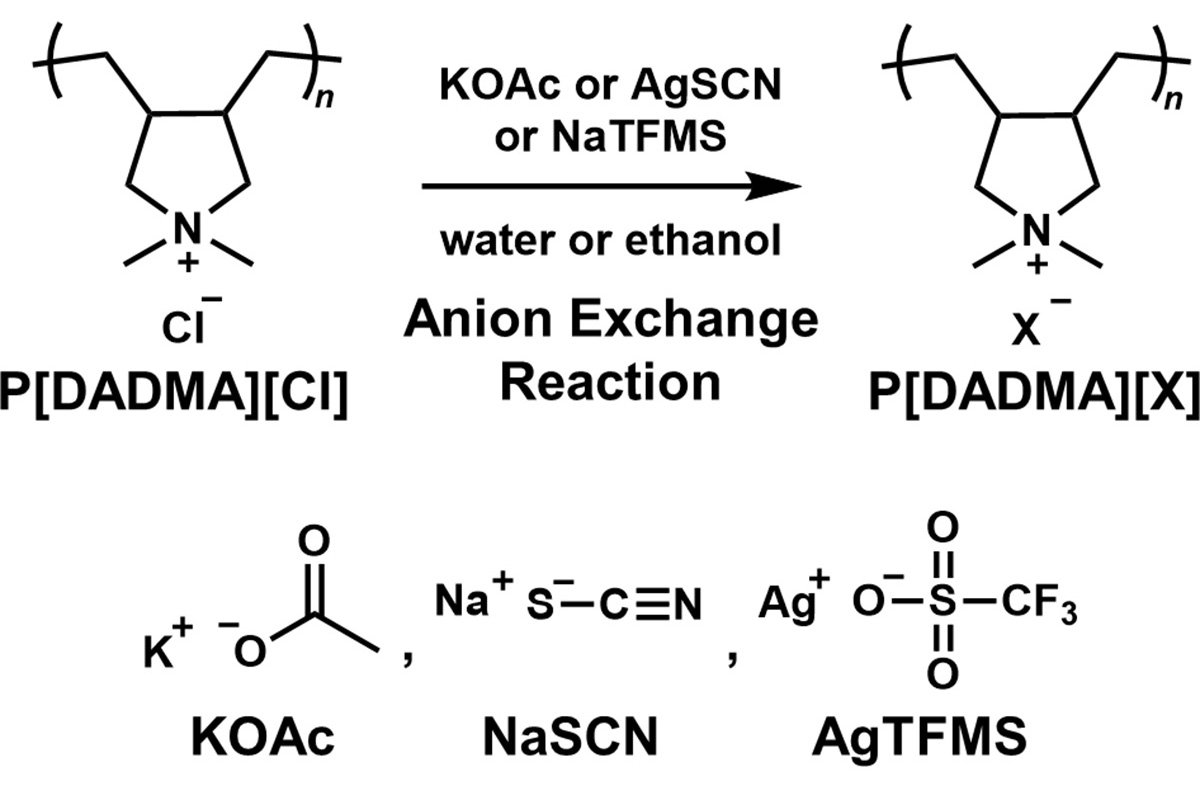
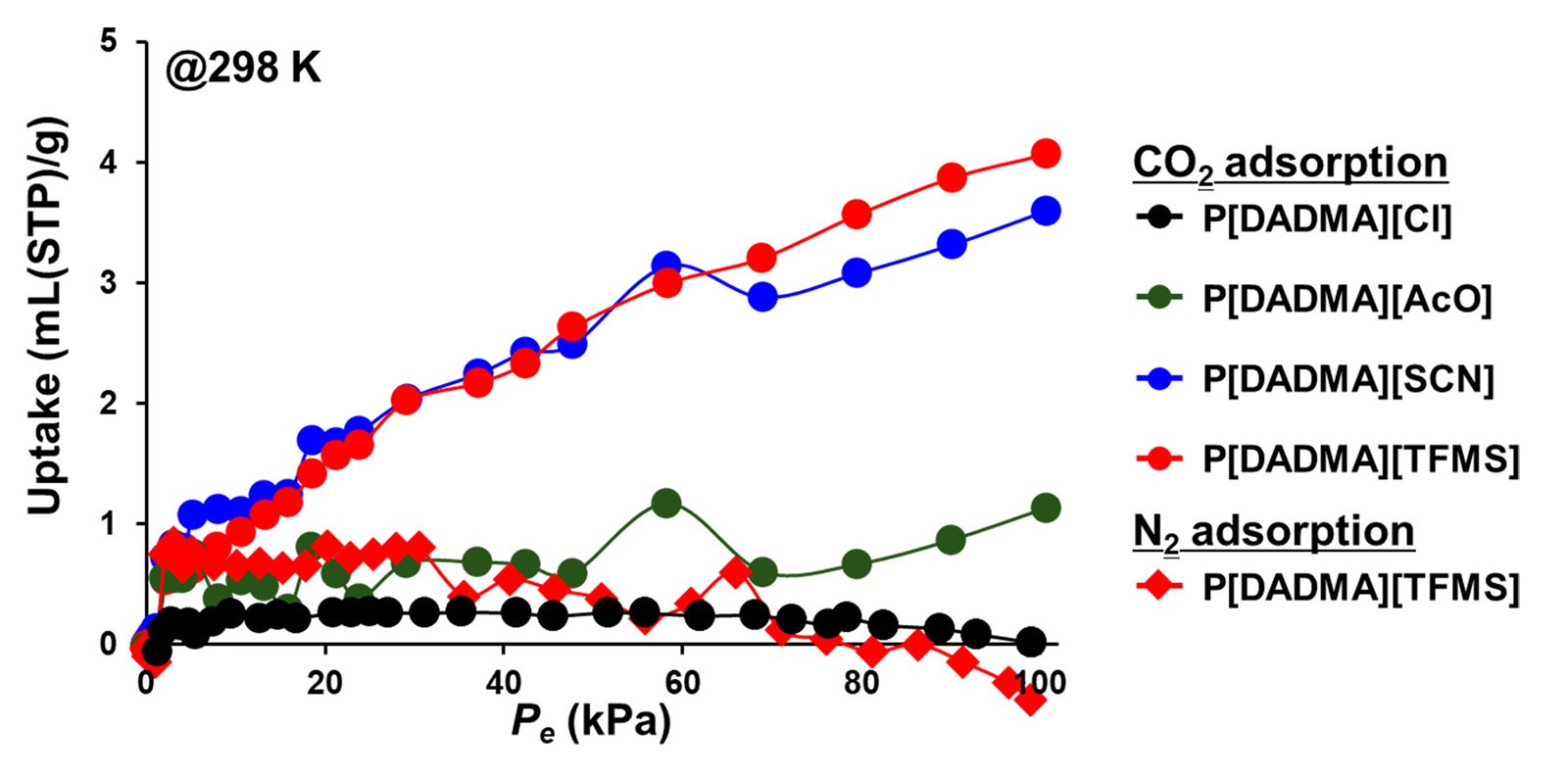
The joint research team--which also includes Kazuhiko Igarashi, Senior Technical Supervising SV at Nittobo--successfully removed inorganic salts by precisely purifying the PILs. They discovered that increasing the size of the counter anion significantly improves the CO2 adsorption capacity. Notably, the material using the largest anion achieved an adsorption capacity seven times greater than the raw material.
Developing efficient ways to capture and separate CO₂ from the atmosphere and industrial emissions is an urgent challenge in addressing global warming. PILs are considered promising materials for this purpose because they combine the high CO₂ affinity of ionic liquids with the stability and ease of processing of polymers. In particular, PILs with a quaternary ammonium structure are known to perform well. However, until now, the effects of residual metal ions from inorganic salts formed during synthesis have not been fully studied.
In this work, the researchers focused on poly(diallyldimethylammonium chloride) (P[DADMA][Cl]), a material with a high density of positive charges. They replaced the chloride (Cl⁻) ion with three anions of different sizes--acetate (AcO⁻), thiocyanate (SCN⁻), and trifluoromethanesulfonate (TFMS⁻)--to examine how anion size affects CO₂ adsorption.
A key achievement was completely removing inorganic salt impurities. The researchers used Scanning Electron Microscopy-Energy Dispersive X-ray Spectroscopy (SEM-EDX) to confirm the total disappearance of chlorine from the raw material and any reaction by-products, ensuring the production of high-purity PILs.
The results clearly showed that CO₂ adsorption increases as the size of the anion increases. The material with the largest anion (TFMS⁻) achieved the highest performance, with an adsorption capacity seven times greater than the starting material.
This research has established a new performance-enhancing approach of "precisely designing the anion size" for PILs. The findings are expected to contribute significantly to the future enhancement of CO2 capture systems and gas separation membranes.

- Publication Details:
Title: Anion size-dependent carbon dioxide adsorption capacity in high-purity diallyldimethylammonium-based poly(ionic liquid)s
Authors: Kohei Okubo, Showa Kitajima, Hitoshi Kasai, Kiyotaka Maruoka, Yuta Takahashi, Yoko Teruuchi, Minoru Takeuchi, Kazuhiko Igarashi and Kouki Oka
Journal: Reaction Chemistry & Engineering
DOI: 10.1039/D5RE00535C






