They say that life comes without an instruction manual, but that's not entirely true. Each cell in our body lives according to instructions issued by its DNA in the form of RNA molecules. RNA was recently thrust into the limelight as the basis of innovative COVID-19 vaccines, but much fundamental knowledge about this vital molecule - for example, how it manages to make its way in the cell to a designated location - is still lacking. Researchers at the Weizmann Institute of Science have now discovered a cellular "postal code" system that makes sure all RNA gets to the right place, right on time.
After the RNAs are produced in the nucleus, some stay there to regulate gene expression, but most - especially those that carry the recipes for proteins - are slated to leave the nucleus for the cytoplasm, where proteins are manufactured. Previous studies aiming to clarify how RNAs get to their assigned locations had produced conflicting results. Some suggested that the routes of the string-like, linear RNA molecules may be dictated by information contained in their loose ends. Yet some RNAs are circular and, obviously, do not have ends. Other studies found hints that certain short segments within RNA molecules could function as zip codes, defining the neighborhood in the cell where each RNA belongs, but different studies reported on different zip codes, and there was limited understanding of how such zip codes might work.

Research student Maya Ron and Prof. Igor Ulitsky, both of the Weizmann Institute of Science's Immunology and Regenerative Biology and Molecular Neuroscience Departments, tested the zip code hypothesis using a technique known as a "massively parallel RNA assay," developed in part in Ulitstky's lab. The technique makes it possible to study thousands of different RNAs simultaneously, obtaining results within days instead of the years it would previously have taken to study these same RNAs one by one. The scientists inserted thousands of different RNA segments into various "host" RNA molecules - linear or circular - copies of which were then introduced into millions of cells. After separating the nucleus from the cytoplasm of these cells, the researchers could tell where their RNAs had ended up.
After investigating some 8,000 genetic segments in this manner, Ron and Ulitsky found that several dozen of them indeed serve as zip codes. These zip codes instruct some RNAs to stay in the nucleus, tell others to move into the cytoplasm immediately, and direct still others to make this move only after lingering in the nucleus for a while. The researchers also discovered several proteins that serve as "postal clerks" whose job it is to bind to RNAs, "read" their zip codes and dispatch the RNAs to the locations encoded there.
""Many companies are now developing RNAs to be used as drugs or vaccines. Understanding how they get to their locations in the cell may help engineer artificial RNAs with desired properties"
Remarkably, there was a clear-cut division between linear and circular RNAs within this "postal system." To begin with, the same zip code could assign an RNA to a different location, depending on whether it was linear or circular. Moreover, two sets of postal clerks ran the sorting, one for the linear RNAs and one for the circular. In fact, each of the clerks issued their own specific kind of instructions. For example, one protein, called IGF2BP1, bound mainly to linear RNAs, promoting their export from the nucleus. Another one, called SRSF1, specialized in directing circular RNAs to stay in the nucleus. When the scientists blocked the activity of individual proteins, the RNAs sorted by each of these postal clerks failed to reach the correct locations in the cell.
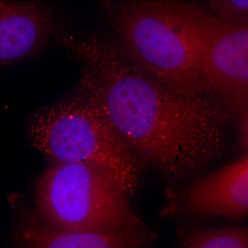
In addition to shedding new light on the functioning of the genome, these findings may prove useful in designing RNA-based therapies. "Many companies are now developing RNAs to be used as drugs or vaccines," Ulitsky says. "Understanding how they get to their locations in the cell may help engineer artificial RNAs with desired properties. For example, if we want an RNA drug to make large amounts of a certain protein, it can be designed to spend most its time in the cytoplasm, where this protein can be produced."
The study's findings may be particularly valuable for the use of circular RNAs, which have become the focus of research relatively recently and which are less well understood than linear RNAs. "In nature, only a small percentage of RNAs are circular, but they are more stable than linear ones and therefore increasingly used in drug design," Ron explains.
Science Numbers
A typical linear protein-producing RNA molecule spends 20% of its existence in the nucleus and 80% in the cytoplasm, where the proteins that it encodes are manufactured.
Prof. Igor Ulitsky's research is suppored by the Gladys Monroy and Larry Marks Center for Brain Disorders, the Helen and Martin Kimmel Institute for Stem Cell Research, the David and Fela Shapell Family Center for Genetic Disorders Research, the Kekst Family Institute for Medical Genetics, Miel de Botton, and Katy and Gary Leff. The Arlyn Imberman Research Fellow Chair supports a staff scientist in Prof. Ulitsky's lab.
Prof. Ulitsky is the head of the Abisch-Frenkel RNA Therapeutics Center.






