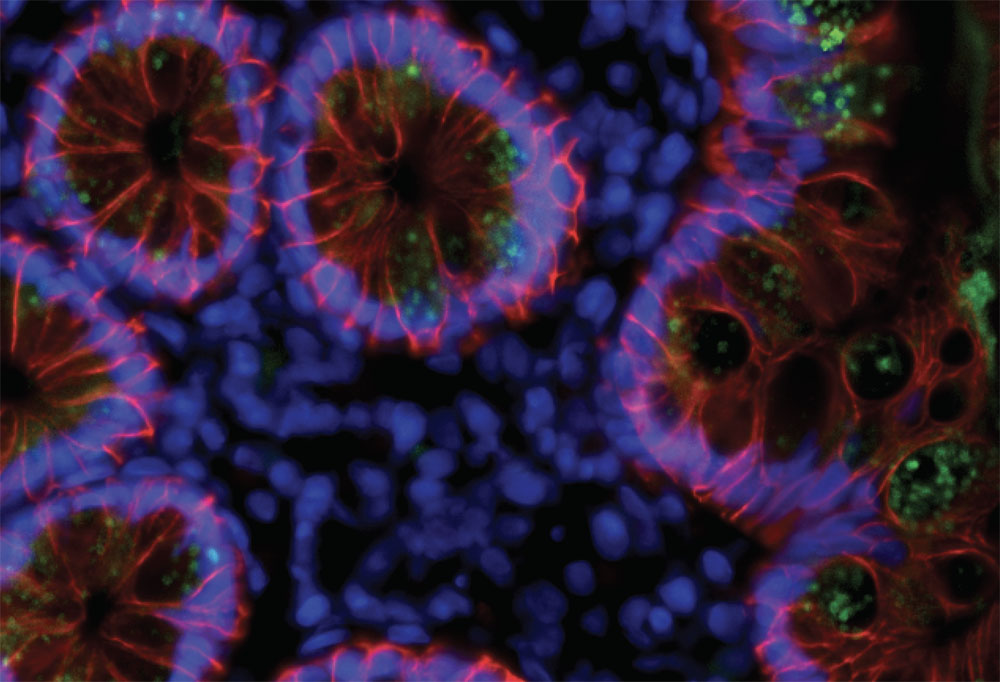
Traces of SARS-CoV-2 (green) in the cells lining people's intestines three months after infection.
As the number of people who have acquired-and then fought off-SARS-CoV-2 climbs ever higher, a critical question has grown in importance: How long does their immunity to the novel coronavirus last? A new Rockefeller study offers an encouraging answer, suggesting that people who recover from COVID-19 are protected against the virus for at least six months, and likely much longer.
The findings, published in Nature, provide the strongest evidence yet that the immune system "remembers" SARS-CoV-2 and, remarkably, continues to improve the quality of antibodies even after the infection has waned. Antibodies produced months after the infection showed increased ability to block the virus, as well as its mutated versions such as the South African variant.
The researchers found that these improved antibodies are produced by immune cells that have kept evolving, apparently due to a continued exposure to the remnants of the virus hidden in the gut tissue.
Based on these findings, researchers suspect that when the recovered patient next encounters the virus, the response would be both faster and more effective, preventing re-infection.
"This is really exciting news. The type of immune response we see here could potentially provide protection for quite some time, by enabling the body to mount a rapid and effective response to the virus upon re-exposure," says Michel C. Nussenzweig, the Zanvil A. Cohn and Ralph M. Steinman Professor and head of the Laboratory of Molecular Immunology, whose team has been tracking and characterizing antibody response in Covid-19 patients since the early days of the pandemic in New York.
Long-lasting memory
Antibodies, which the body creates in response to infection, linger in the blood plasma for several weeks or months, but their levels significantly drop with time. The immune system has a more efficient way of dealing with pathogens: instead of producing antibodies all the time, it creates memory B cells that recognize the pathogen, and can quickly unleash a new round of antibodies when they encounter it a second time.
But how well this memory works depends on the pathogen. To understand the case with SARS-CoV-2, Nussenzweig and his colleagues studied the antibody responses of 87 individuals at two timepoints: one month after infection, and then again six months later. As expected, they found although antibodies were still detectable, their numbers had markedly decreased by the six-month point. Lab experiments showed that the ability of the participants' plasma samples to neutralize the virus was reduced by five-fold.
In contrast, the patients' memory B cells, specifically those that produce antibodies against SARS-CoV-2, did not decline in number, and even slightly increased in some cases. "The overall numbers of memory B cells that produced antibodies attacking the Achilles heel of the virus, known as the receptor-binding domain, stayed the same," says Christian Gaebler, a physician and immunologist in Nussenzweig's lab. "That's good news because those are the ones that you need if you encounter the virus again."
Viral stowaways
A closer look at the memory B cells revealed something surprising: these cells had gone through numerous rounds of mutation even after the infection resolved, and as a result the antibodies they produced were much more effective than the originals. Subsequent lab experiments showed this new set of antibodies were better able to latch on tightly to the virus and could recognize even mutated versions of it.
"We were surprised to see the memory B cells had kept evolving during this time," Nussenzweig says. "That often happens in chronic infections, like HIV or herpes, where the virus lingers in the body. But we weren't expecting to see it with SARS-CoV-2, which is thought to leave the body after infection has resolved."
SARS-CoV-2 replicates in certain cells in the lungs, upper throat, and small intestine, and residual viral particles hiding within these tissues could be driving the evolution of memory cells. To look into this hypothesis, the researchers teamed up with Saurabh Mehandru, a former Rockefeller scientist and a physician at Mount Sinai Hospital, who had been examining biopsies of intestinal tissue from people who had recovered from COVID-19 on average three months earlier.
In seven of the 14 individuals studied, tests showed the presence of SARS-CoV-2's genetic material and its proteins in the cells that line the intestines. The researchers don't know whether these viral left-overs are still infectious or are simply the remains of dead viruses.
The team plans to study more people to better understand what role the viral stowaways may play in both the progression of the disease and in immunity.






